Ultrasonic Study of Thermophysical Characteristics of Amino acids in Aqueous 2% D-glucose Solution at Varied Temperatures
DOI:
https://doi.org/10.61343/jcm.v3i02.160Keywords:
Hydrophilic, Hydrophobic, volumetric studies, electrostriction effect, apparent molar volume, partial molar volumeAbstract
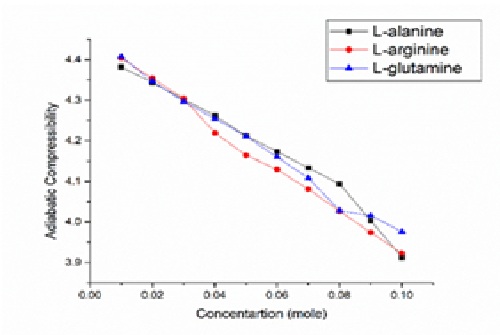
Thermodynamic parameters like density, viscosity, and ultrasonic velocity have been measured for 0.01 to 0.1 molal concentration of L-alanine, L-arginine and, L-glutamine in 2% aqueous D-glucose solution at different temperature ranges from 293.15 to 313.15K. These measurements have been performed to evaluate some important parameters like adiabatic compressibility, molar hydration number, apparent molar volume, partial molar volume, apparent molar compressibility, limiting apparent molar compressibility, and their constant and viscosity A and B coefficients of the Jones-Dole equation. These parameters have been interpreted as molecular interactions in terms of solute-consolute and solute-solvent interactions.
References
T. Gaudin, P. Rotureau, I. Pezron, G. Fayet, Comput. Theor. Chem., 1101 (2017), 20-29.
B. Lu, M. Vayssade, Y. Miao, V. Chagnault, E. Grand, A. Wadouachi, D. Postel, A. Drelich, C. Egles, I. Pezron, Colloids Surf. B: Biointerfaces, 145 (2016), 79-86.
K. Thalberg, B. Lindman, in: E. D. Goddard, K. P. Ananthapadmanabhan (Eds.), “Interactions of Surfactants with Polymers and Proteins”, CRC Press, Bocca Raton, FL, 1993.
C. E. Forney, C. E. Glatz, Biotechnol. Prog. 11 (1995), 260-264.
S. K. Singh, A. Kundu, N. Kishore, J. Chem. Thermodyn. 36 (2004), 7-16.
T. Cserháti, Environ. Health Perspect. 103 (1995) 358.
S. Chauhan, J. Jyoti, G. Kumar, J. Mol. Liq., 159 (2011), 196-200.
Z. Yan, X. Wang, R. Xing, J. Wang, J. Chem. Eng. Data, 54 (2009), 1787-1792.
R. Curtis, J. Prausnitz, H. Blanch, Biotechnol. Bioeng., 57 (1998), 11–21.
M. Singh, M. Pandey, R. K. Yadav, H. Verma, J. Mol. Liq., 135 (2007), 42–45.
Gekko, K. J. Biochem. 1981, 90, 1633-1641.
Gekko, K. J. Biochem. 1981, 90, 1633-1652.
Palecz, B., Piekarski, H. J. Solution chem. 1997, 26, 621-629.
S. A. Lippard, J. M. Berg, “Principles of Bioinorganic Chemistry”, University Science. Books, Mill Valley, CA, 1994.
Z. Yan, X. Wang, R. Xing, J. Wang, J. Chem. Eng. Data, 54 (2009), 1787-1792.
Bhat, R. and J. C. Abluwalia, 1985. “Partial molar heat capacities and volumes of transfer of some amino acids and peptides from water to aqueous sodium chloride solutions at 298.15 K”, J. Phys. Chem, 89: 1099-1105.
Chauhan, M. S., K. Sharma and G. Kumar, 2002. “Transport studies of ion-solvent interactions in acetonitrile-rich regions of benzene and ethanol”, Ind. J. Chem, 40; 481-483.
Jones, G. and M. Dole. 1929. “The viscosity of aqueous solutions of strong electrolytes with special reference to barium chloride”, J. Am. Chem. Soc., 51; 2950-2964.
Chauhan, S., Singh, K., Kumar, K., Neelakantan, S. C., Kumar, G.: “Drug-amino acid interactions in aqueous medium: volumetric, compressibility, and viscometric studies”, J. Chem. Eng. Data, 61, 788-796 (2016).
A. Buranwoski, J. Glinski, “Hydration Numbers of Nonelectrolytes from Acoustic Methods”, Chem. Rev. 2012, 112, 4, 2059–2081.
D. Brahman, B. Sinha, “Partial molar volumes and viscosity B-coefficients of N, N′-ethylene bis (salicylideneiminato) cobalt (II) in binary mixtures of 1,4-dioxane+methanol at T= (298.15, 303.15, 308.15, and 313.15) K”, J. Chem. Eng. Data 56 (2011) 3073–3082.
J. Krakowiak, “Apparent molar volumes and compressibilities of tetrabutyl-ammonium bromide in organic solvents”, J. Chem. Thermodyn. 43 (2011) 882–894.
D. Brahman, B. Sinha, “Partial molar volumes and viscosity B-coefficients for N, N/-ethylene bis(salicylideneiminato)-diaquochromium(III) chloride in methanolic solutions of 1-butyl-2,3 dimethylimidazolium tetrafluoroborate at T= (298.15, 308.15, and 318.15) K”, J. Chem. Thermodyn, 68 (2014) 260–269.
H. Kumar, I. Behal, M. Singla, J. Chem. Thermodyn, 95 (2016) 1-14.
A. K. Nain, M. Lather, J. Chem. Thermodyn., 2016, 102, 22 38, doi: 10.1016/j.jct.2016.06.030.
M. A. Jamal, B. Naseem, S. Naaz, I. Arif, M. Saeed, S. Atiq, “Thermo-acoustic properties of maltose in aqueous amino acids system”, Journal of Molecular Liquids, 309, 112932 (2020).
Mohd. R. and E. Verrall, “Apparent molar volume and adiabatic compressibility studies of aqueous solutions of some drug compounds at 25°C”, Can. J. Chem. 67, 727 (1989).
Prakash S., F. M. Ichhaporia and J. D. Panday, 1964, “Standard study of complex Barium citrate by Ultrasonic waves”, J. Phys., Chem. USA 58:3078.
Downloads
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2025 S. G. Rathod, A. A. Mistry, Rupali Thete, S. A. Shah

This work is licensed under a Creative Commons Attribution 4.0 International License.
Copyright© by the author(s). Published by journal of Condensed Matter. This is an open access article distributed under the terms of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.









