Nanoscale Wettability of Water-Alcohol Mixtures on Graphite Surface: Molecular Dynamics Study
DOI:
https://doi.org/10.61343/jcm.v3i01.124Keywords:
Wetting, Contact Angle, Molecular Dynamics Simulation, Binary MixturesAbstract
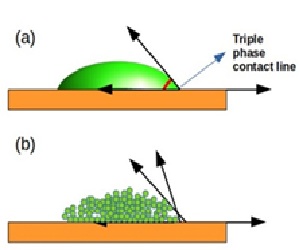
The ability to predict and control surface wettability at the nanoscale is crucial and rapidly evolving, particularly in the fields of microfluidics and nanotechnology. Typically, the contact angle is measured in simulations by creating a liquid droplet on a solid surface, fitting a curve to the droplet’s interface, and determining the angle at the triple-phase contact line. However, at the molecular level, the triple-phase contact line becomes ambiguous due to the continuous motion of molecules, making conventional measurements difficult. This study reports the molecular dynamics simulations to determine the contact angle of water mixed with four primary alcohols (RN-OH, where R = (CNH2N+1), N varies from 1 to 4) on a graphite substrate, using the Hautman and Klein method, which links the microscopic contact angle to the droplet’s average center of mass height through the concepts of equivalent contact area and volume. The water concentration is varied from 50% to 90% in the alcohol mixture. ContactAngleCalculator code is modified to calculate the contact angle of binary liquids. Results show that increasing alcohol concentration leads to greater spreading.
References
S. Damiati, U. B. Kompella, S. A. Damiati, and R. Kodzius, “Microfluidic Devices for Drug Delivery Systems and Drug Screening”, in Genes. 9- 21-103, Feb. 2018.
Y Liu and X Jiang. Lab. Chip, 17(23):3960–3978, 2017.
K Velmurugan, M B Kulkarni, I Gupta, R Das, S Goel, and J Nirmal. Microfluidics and Multi Organs on Chip, pages 107–133, 2022.
A P Thompson et al. Comput. Phys. Commun., 271:108171, 2022.
Development and Testing of the OPLS All-Atom Force Field on Conformational Energetics and Properties of Organic Liquids. J. Am. Chem. Soc., 118(45), 1996.
L S Dodda, I Cabeza de Vaca, J Tirado-Rives, and W L Jorgensen. Nucleic Acids Res., 45(W1), 2017.
J Zielkiewicz. J. Chem. Phys., 123(10):104501, 2005.
F Taherian et al. Langmuir, 29(5):1457–1465, 2013.
W Humphrey, A Dalke, and K Schulten. J. Mol. Graph., 14(1):33–38, 1996.
U Abdulkareem, T R Kartha, and V Madhurima. J. Mol. Model., 28(12):382, 2022.
U Abdulkareem, T R Kartha, and V Madhurima. J. Mol. Model., 29(5):151, 2023.
Y Wang, A Kiziltas, P Blanchard, and T R Walsh. J. Chem. Inf. Model., 62(24):6302–6308, 2022.
C F Fan and T CaÇgin. J. Chem. Phys., 103(20):9053–9061, 1995.
Downloads
Published
How to Cite
Issue
Section
Categories
License
Copyright (c) 2025 Abdulkareem U, V Madhurima

This work is licensed under a Creative Commons Attribution 4.0 International License.
Copyright© by the author(s). Published by journal of Condensed Matter. This is an open access article distributed under the terms of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.









